Pharmaceuticals
Tackling the Unmet Medical Needs
The Otsuka group aims to be an indispensable contributor to people’s health worldwide through two core businesses under the corporate philosophy "Otsuka-people creating new products for better health worldwide." These businesses are the Pharmaceutical Business, which provides comprehensive health support from diagnosis to treatment of diseases, and the Nutraceutical Business*, which helps people maintain and improve daily health. The group strives to pursue new values by contributing to the overall well-being of society through unique solutions as the social environment continues to change. In the Pharmaceutical Business, the group focuses on "unmet medical needs," prioritizing psychiatry and neurology, oncology, and cardiovascular and renal areas. Our goal is to provide comprehensive healthcare services, from the development of therapeutic drugs to diagnostic support, raising awareness, and support for recovery.
- *Nutraceuticals are products that have scientific evidence to be beneficial for maintaining and improving daily health, created from the combination of "Nutrition" and "Pharmaceuticals."
Otsuka group's business strategy
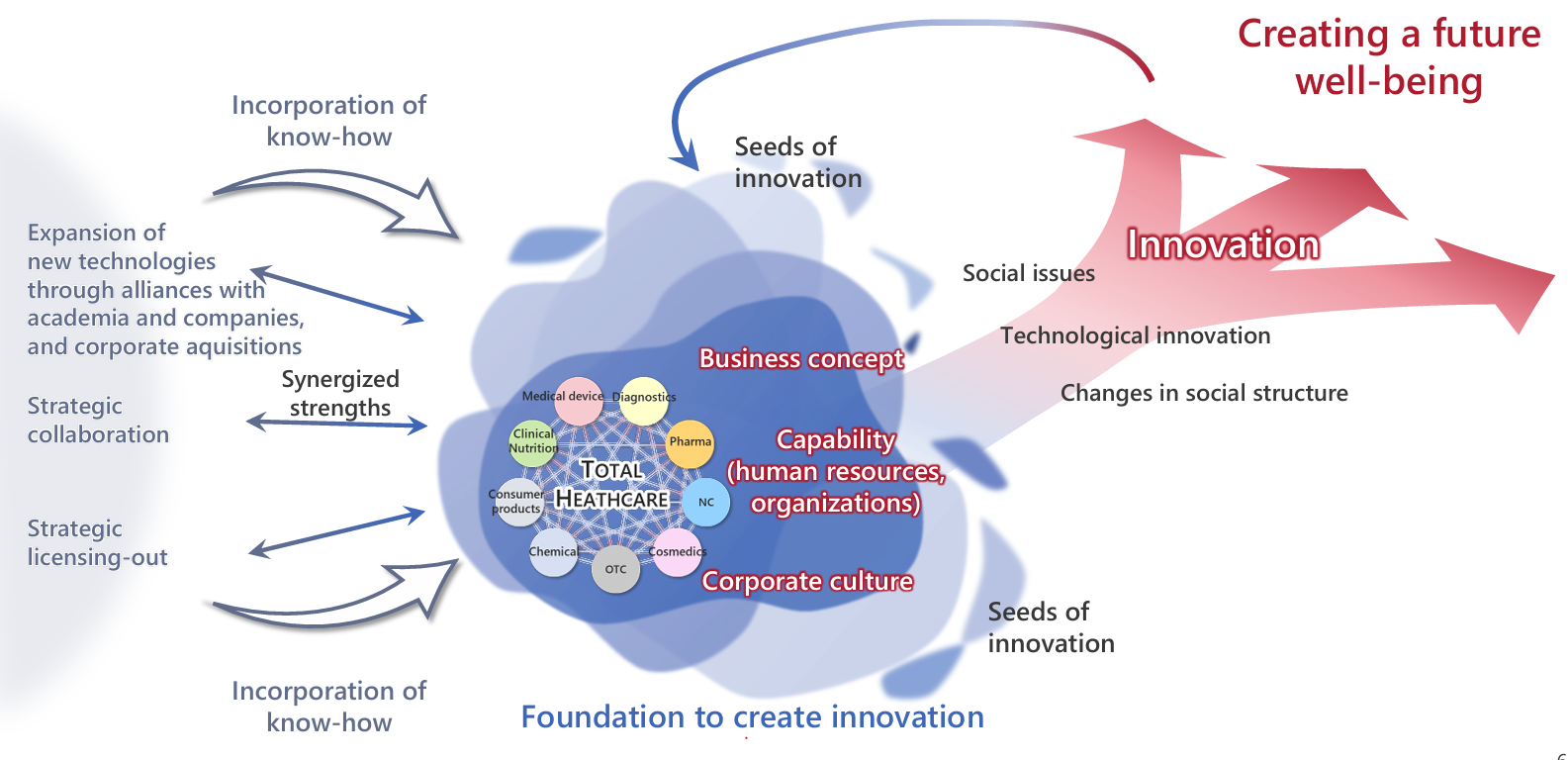
Our Businesses > Our Four Businesses and R&D > Pharmaceuticals
Efforts to Deliver Medicines to More Patients (Access to Medicine)
Enhancing Access to Medicines
The Otsuka group is engaged in the research and development of new drugs and infusions to enhance access to medicines, while also supporting individuals whose access is limited due to economic or other reasons. For example, Otsuka Pharmaceutical has established the Otsuka Global Patient Access Support Policy to enhance patient access to treatment. Specifically, in Asian countries and regions, it is implementing the Otsuka Patient Assistance Program using the leukemia treatment drug providing financial support such as drug provision to patients who cannot afford treatment costs due to insufficient insurance reimbursement or economic reasons.
Efforts to Expand Access to the Tuberculosis Drug
Tuberculosis is one of the world's three major infectious diseases, along with AIDS and malaria. In 2023, an estimated 10.8 million people worldwide were infected, and 1.25 million died from it, making it the leading cause of death from infectious diseases (estimated by WHO, 2023). Otsuka Pharmaceutical is working to expand access to its proprietary multidrug-resistant tuberculosis treatment drug, to more patients in to order eradicate tuberculosis. By collaborating with organizations such as the “Stop TB Partnership's Global Drug Facility (GDF)”, which aims for sustainable procurement in developing countries with many patients, such as Africa and Asia, Otsuka Pharmaceutical is ensuring this drug available in nearly all countries where it is needed.. Since 2014, it has delivered over 120,000 cases for treatment to countries working to control multidrug-resistant tuberculosis. It is also currently developing a successor tuberculosis treatment drug.
Otsuka Pharmaceutical > Unmet medical needs
Stable Supply of Infusions at Appropriate Prices
The Otsuka group is also engaged in efforts to improve the medical environment surrounding the provision of pharmaceuticals at appropriate prices. For example, infusions are highly necessary for medical care and need to be continuously and stably supplied to the market. Therefore, Otsuka Pharmaceutical Factory, Inc. manufactures infusions locally, contributing to stable supply, appropriate pricing in each country, and job creation, thereby contributing to local communities.
Efforts to Expand Access to Investigational Drugs (Expanded Access Program)
The Otsuka group, through the Expanded Access Program, allows the provision of investigational drugs for patients who cannot expect sufficient efficacy from existing treatments and cannot participate in clinical trials due to their disease condition. These drugs are provided to patients who do not meet the criteria for clinical trial participation, based on physician applications and meeting specified conditions. This program thereby expands access to investigational drugs.
Otsuka Pharmaceutical D&C (U.S.) Initiatives
Taiho Oncology (U.S.) Initiatives

In addition, when starting treatment in the U.S., Taiho Oncology, Inc. provides services such as insurance verification, financial assistance for treatment costs, and personalized nursing support to patients, caregivers, and healthcare professionals.
Patient Support Programs
In some countries and regions, Taiho Oncology offers various programs to support patients and caregivers when purchasing specific Otsuka group products.
Otsuka Pharmaceutical (U.S.)
The Otsuka Patient Assistance Foundation, Inc. (OPAF)
The Otsuka Patient Assistance Foundation, Inc. (OPAF) is a non-profit organization that provides Otsuka's treatment drugs free of charge to uninsured patients or those whose insurance does not sufficiently cover treatment costs.

(U.S.) Otsuka America Pharmaceutical, Inc. (OAPI)

Otsuka Patient Support™ combines resources, tools, and support teams for patients, caregivers, and healthcare professionals to support patients.
Taiho Parma Canada Inc.

Taiho Pharma Canada, Inc. offers personalized patient support program (Conexus Patient support Program™) that includes guidance on medical expense reimbursement, one-on-one support from dedicated nurses, specialized pharmacy services, home delivery services, and educational services for patients, caregivers, and healthcare professionals when starting treatment.
