Procurement
Core Principles
The Otsuka group, as a total healthcare company, is committed to providing a stable supply of safe and reliable products that are trusted by all. In our group's procurement activities, we consider human rights, working conditions, anti-corruption, and the environment, in addition to quality, safety, and stable supply. We aim to implement sustainable procurement with our business partners, and strive to build a sustainable supply chain and improve the corporate value of both parties.
The Otsuka Group's Materiality and Related SDGs

- Create More Sustainable Future
with Business Partners
Social Issues
- Destabilization such as pandemics, geopolitical risks of raw material procurement and product supply due to supply chain disruption
- Building a sustainable supply chain
Related SDGs

| Strategy | Measures | Indicators |
|---|---|---|
|
|
|
|
|
|
Sustainable Procurements With Our Suppliers
In March 2024, the Otsuka group established the Otsuka Group Business Partner Code of Ethics to express our intention to conduct corporate activities based on high ethical standards in cooperation with our business partners.
In procurement activities, the group has established the Otsuka Group Procurement Policy and the Otsuka Group Sustainable Procurement Guidelines to guide the group’s actions and to communicate these guidelines to suppliers.
In November 2024, the group also established a Speak Up Line for its business partners, including suppliers, to consult and report any suspected misconduct or violations of laws and regulations by individuals associated with the group.
To deliver high-quality and sustainable products to patients and consumers, the group collaborates with its suppliers not only to ensure quality, safety, and stable supply, but also to promote ethical and sustainable procurement practices. Through these efforts, the group aims to contribute to a sustainable society and enhance corporate value for both parties.
Otsuka Group Procurement Policy
Download our procurement policy here.
Otsuka Group Sustainable Procurement Guidelines
Download our procurement guidelines here.
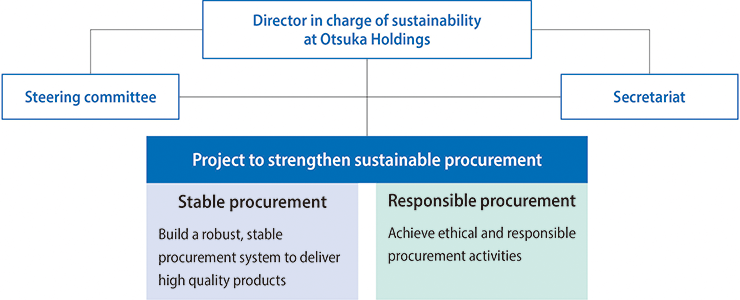
Promotion System
A Sustainable Procurement Project,* led by the director in charge of sustainability at Otsuka Holdings, was launched in 2022 and its progress and issues are shared every quarter.
This project promotes activities with two pillars: “Stable Procurement,” aiming to build a solid procurement system to deliver high-quality products, and “Responsible Procurement,” aiming for ethical and sustainable procurement activities.
*Participants include directors and personnel in charge of procurement, production, IT, and compliance at Otsuka Holdings, Otsuka Pharmaceutical, Otsuka Pharmaceutical Factory, Taiho Pharmaceutical, Otsuka Warehouse, Otsuka Chemical, Otsuka Foods, Otsuka Medical Devices, EN Otsuka Pharmaceutical, Otsuka Techno, Okayama Taiho Pharmaceutical, Otsuka Packaging Industries, and JIMRO
Promotion Structure of the Sustainable Procurement Project
Approach
Building Partnerships with Supplier Companies
The group endorses the aims of the Council on Promoting Partnership Building for Cultivating the Future, which is promoted by the Cabinet Office and the Small and Medium Enterprise Agency (SMEA) and has published a Declaration of Partnership Building to create relationships of trust and further strengthen cooperation with supplier companies.
Currently, nine companies have made the declaration: Otsuka Holdings, Otsuka Pharmaceutical, Otsuka Pharmaceutical Factory, Taiho Pharmaceutical, Otsuka Foods, Otsuka Chemical, Otsuka Packaging, EN Otsuka Pharmaceutical, and Otsuka Techno.
Implementing Supplier Due Diligence
To ensure a stable product supply, the group has conducted due diligence in new suppliers and risk assessments of key raw materials. The group has also implemented risk countermeasures for raw material procurement, such as identifying potential risks and sourcing from multiple suppliers.
While some of the group’s overseas and domestic operating companies already evaluate new suppliers based on human rights, labor conditions, anti-corruption measures, and environmental impact, expanding this evaluation to all group companies remains a challenge for the future.
Stable Procurement
To enhance its capability to deliver high‑quality products and to build a stable procurement framework, the Otsuka group established a cross‑functional working team.
In 2024, it developed a platform to centrally manage procurement information that had previously been managed separately by each operating company. As it is essential for the group to share its purpose and significance with its business partners when operating this platform, the group held briefing sessions for direct‑material* suppliers in Japan and advanced the development of an integrated information‑sharing framework.
In 2025, approximately 80% of the targeted suppliers participated in and cooperated with this initiative, enabling the group to obtain their consent and proceed with the registration of site information. Moreover, in the same year, the group utilized the platform during an actual disaster to conduct a supplier status survey. As a result, it achieved a 100% response rate during the incident, enabling far more efficient information gathering compared with conventional individual confirmations. The group will continue to improve the speed of assessing supply chain impacts during disasters and strengthen its initial response capabilities, as we work to further expand the operation of this platform.
*Raw materials and components used directly in the manufacture of a product. This designation includes materials that are part of the product and key materials consumed in the manufacturing process.
Message from the Working Group on Stable Procurement

Daisuke Yagi
Director, Purchasing Department
Otsuka Pharmaceutical Co., Ltd.
Globalization and the increased complexity of supply chains have accelerated in recent years. In addition, there is a global need to respond to risks such as natural disasters, pandemics, and geopolitical risks, as well as to address sustainability issues that take human rights and social concerns into account.
In this context, the group believes that visualization of the supply chain, centralized management of information, and close information sharing with suppliers will enable us to provide a stable supply to patients, medical professionals, and consumers. With this in mind, Otsuka Holdings and its five operating companies* have introduced a common platform for supply chain information management in 2024.
In normal times, the group will identify high-risk items to mitigate potential risks in advance, and in emergencies, the group will ensure an efficient initial response through prompt information sharing with suppliers to realize an even stronger and more stable supply system.
*Otsuka Pharmaceutical, Otsuka Pharmaceutical Factory, Taiho Pharmaceutical, Otsuka Foods, and Otsuka Chemical
Responsible Procurement
To promote ethical, sustainable, and responsible procurement practices, the group operates a cross‑functional supplier risk management program through a working group composed of 12 affiliates*. This working group ensures that all suppliers are informed of and agree to the “Otsuka Group Procurement Policy” and the “Otsuka Group Sustainable Procurement Guidelines.”
In addition, supplier assessments are conducted in stages by combining evaluations through EcoVadis, which is an internationally recognized assessment platform, with the SAQ (Self‑Assessment Questionnaire) administered by our group.
Indicator: Number of communication engagements with suppliers
(Assessment scope: Approximately 620 direct material suppliers in Japan)
・Phase 1 (2025): Completed for 389 suppliers
・Phase 2 (2026): Approximately 170 suppliers (planned)
・Phase 3 (2027): Approximately 60 suppliers (planned)
Based on the results of the Phase 1 assessment, seven suppliers were identified as potentially exposed to risks. The working group conducted interviews with these suppliers based on our corrective action manual and confirmed that none of the concerns were substantiated.
In addition, the group appropriately responds to supplier assessments requested by business partners and aims to contribute to enhancing the sustainability of the entire global supply chain, including through improvements achieved via dialogue and engagement.
Going forward, the group will expand this program to overseas group companies and reinforce local risk management and responsible procurement initiatives.
Message from the Working Group on Responsible Procurement

Shingo Takida
Manager, Purchasing Department
Otsuka Pharmaceutical Factory, Inc.
Our subcommittee is dedicated to promoting ethical and sustainable sourcing, alongside ensuring the stable procurement of raw materials and product supply. Since May 2025, we have been working with Otsuka Holdings and 12 affiliates* to assess suppliers.
Due to the large number and diversity of suppliers, it is difficult to expect a uniform response. Consequently, we often address individual issues, guided by the spirit of Ryukan-godo—sharing common values with our suppliers and working together toward a cooperative approach.
We will continue enhancing the sustainability of our supply chain to help realize a sustainable society and increase corporate value.
*Otsuka Pharmaceutical, Otsuka Pharmaceutical Factory, Taiho Pharmaceutical, Otsuka Warehouse, Otsuka Chemical, Otsuka Foods, Otsuka Medical Devices, EN Otsuka Pharmaceutical, Otsuka Techno, Okayama Taiho Pharmaceutical, Otsuka Packaging Industries, and JIMRO
Message from Pharmavite

Ray Gosselin
EVP, Chief Operations Officer
Pharmavite, LLC
As a leader in the health and wellness industry with a commitment to responsibly produce the highest-quality products for our consumers, it is essential to Pharmavite that we continuously improve our efforts in sustainability. In my role as Chief Operations Officer at Pharmavite, I am charged with ensuring that our suppliers share our commitment to sustainability and understand the urgency of the issue.
The first step in our sustainability agenda is to gain transparency with our suppliers and their suppliers' sustainability performance, and taking proactive steps to do so reflects the important values of our organization: Do The Right Thing, Care Deeply, and Do Something. To that end, Pharmavite conducts individual sustainability performance assessments of our supply chain partners using EcoVadis, a global ESG rating company based on international standards. This assessment is a key factor for Pharmavite sourcing and supplier review as we believe this process is critical to our supply chain sustainability management.
We believe that this strategic initiative with our supply chain partners will enhance our mutual corporate values and pave the way for a more sustainable future.
Robust Supply Chain Network for the Stable Supply of Prescription Drugs
Otsuka Pharmaceutical established the Global Supply Chain Committee (GSCC) in 2017 to make decisions on important production and supply-related issues for anticancer drugs, orphan drugs, tuberculosis drugs and others to ensure a stable supply of pharmaceuticals in the global market. Otsuka Pharmaceutical and our overseas subsidiaries are cooperating to strengthen the supply chain. This includes the addition of manufacturing bases in 2022 to bolster the supply chain in the global market for core products.
